|
1/20/2024 0 Comments Magnesium atomic mass 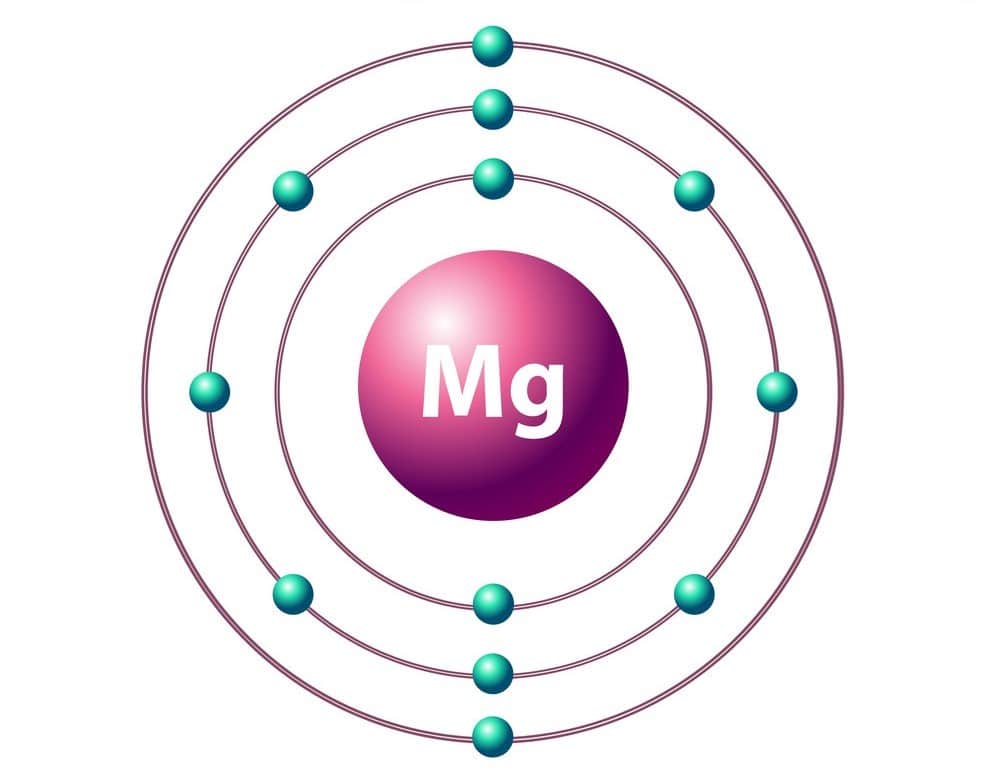
But care must be taken while solving the problem.Atomic mass of all elements (along with the rounded off values) is mentioned in the chart below. Here, there is no interference of units as everything is in the form of percentages. General concept of average atomic mass helps us reach the answer. 
Isotope of an element have the same atomic number but the number of neutrons vary in their nucleus thus, atomic masses are different.Īverage atomic mass of magnesium = 24.31 amu Such elements reside on the same position in the periodic table where the most abundant form of isotope is placed. The elements in the modern periodic table can exist in their isotopic form in actual nature where the elements have the same atomic number but different atomic mass. The atomic mass of any element is empirical and is based on the number of protons in its nucleus purely. Let us know the basic concept of average atomic mass Its unit is generally expressed as amu (Dalton). The average atomic mass of an element is the sum of product of the masses of its isotopes to its abundance in nature. 
Also, only one option can be correct while we solve this question. We need to know the basics of this concept to solve this illustration. 
Hint: An average atomic mass plays an important role while solving such problems. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
